Valence electrons are an essential concept in chemistry that plays a crucial role in understanding the behavior and properties of elements. Elements with fewer valence electrons tend to be more reactive as they strive to achieve a full outer shell, while elements with a full outer shell are typically stable and less reactive. The number and arrangement of valence electrons impact an element’s reactivity. The reactivity of elements is influenced by their valence electrons. This process leads to the formation of stable compounds with lower energy compared to individual atoms. Valence electrons are involved in the formation of chemical compounds.Ĭhemical compounds are formed through the sharing or transfer of valence electrons between atoms. For example, ions strive to achieve a stable electron configuration by gaining or losing enough electrons to attain a full outer shell. The resulting number of valence electrons in the ion affects its stability. When atoms gain or lose valence electrons, they form ions. Valence electrons determine the stability of ions. These mobile valence electrons are responsible for the high electrical and thermal conductivity observed in metals. In metallic elements, valence electrons are delocalized and free to move throughout the metal lattice. Valence electrons are responsible for the conductivity of metals.

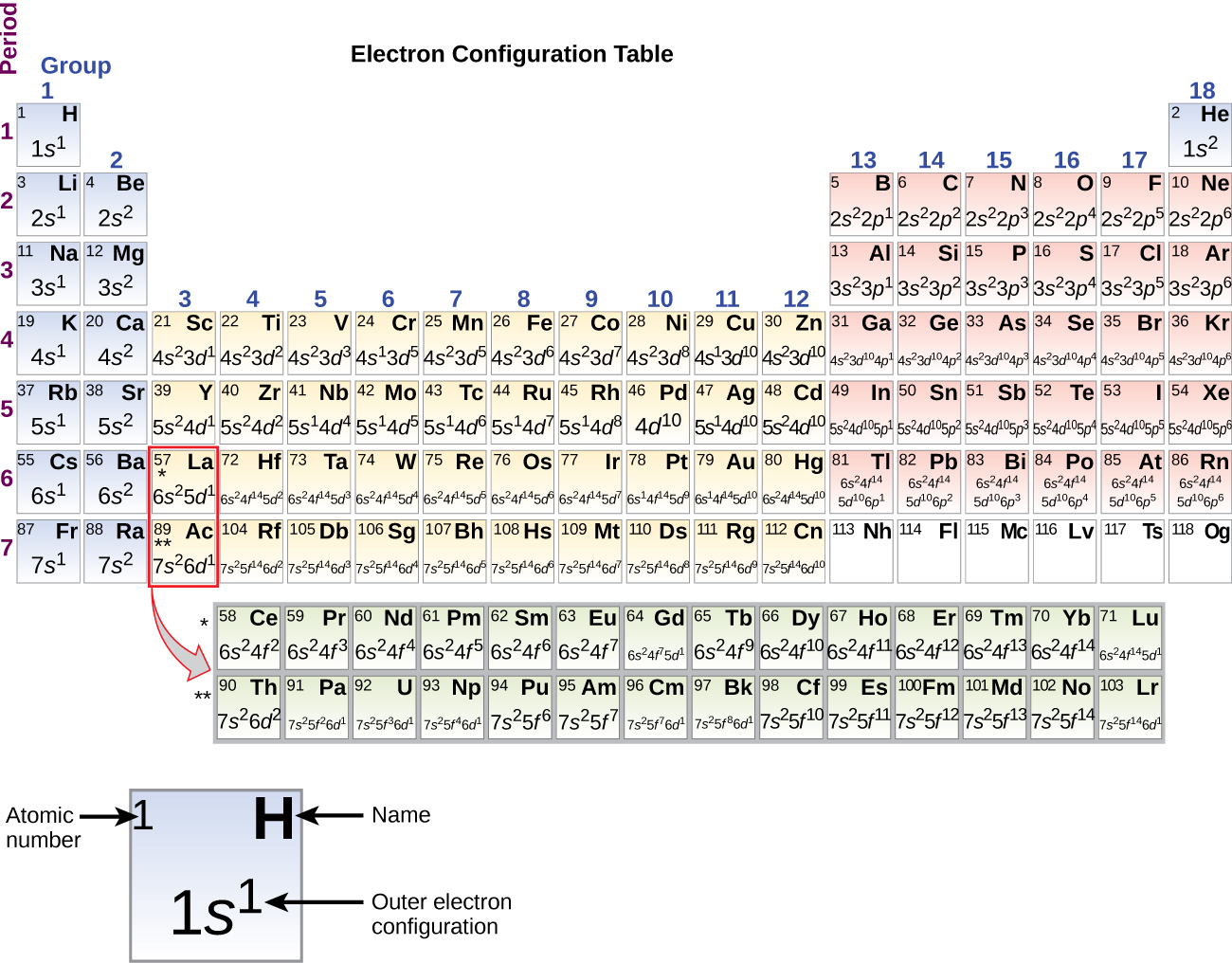
This rule helps to explain the formation of chemical bonds in many compounds. The octet rule states that atoms tend to gain, lose, or share valence electrons to achieve a stable electron configuration with eight valence electrons. Read also: 20 Astonishing Facts About Cosmic Ray Composition The octet rule guides valence electron configuration. For example, elements in Group 1 have one valence electron, while elements in Group 18 have eight valence electrons. The group number of an element in the periodic table represents the number of valence electrons it possesses. Valence electrons can be identified using the periodic table. Elements with the same number of valence electrons exhibit similar chemical behavior. The number of valence electrons in an atom determines its group and period in the periodic table. Valence electrons determine an element’s placement in the periodic table. They are involved in the formation of chemical bonds between atoms, which determines the reactivity and properties of elements. Valence electrons are the electrons located in the outermost shell of an atom. For s-block and p-block elements, the valence electron is equal to the group number of the element.Valence electrons play a crucial role in chemical bonding.

For calculating the valence electron we need to look at the groups in the periodic table.Periods are horizontal rows of the periodic table, while groups are vertical columns (down) the table. Groups and periods are two ways of categorizing elements in the periodic table.Periodic table is a table listing the chemical elements in order of their atomic numbers.from the periodic table or from the electronic configuration. There are two ways to find valence electrons i.e.An electron that is part of an atom's outer shell and can take part in the formation of a chemical bond if the outer shell is not closed is known as a valence electron.JEE Main 2022 Question Paper Live Discussion.Difference Between Selling And Marketing.TS Grewal Solutions Class 11 Accountancy.TS Grewal Solutions Class 12 Accountancy.CBSE Previous Year Question Papers Class 12.CBSE Previous Year Question Papers Class 10.NCERT Solutions For Class 6 Social Science.NCERT Solutions for Class 7 Social Science.NCERT Solutions for Class 8 Social Science.
